![]()
On March 20, 2026, a single patent expired quietly in India. No press conference. No breaking news. Just a lock falling off a door — and 40+ companies charging straight through it.
The drug? Semaglutide. You know it as Ozempic — the celebrity weight-loss injection that turned Novo Nordisk into a $570 billion company and made Eli Lilly the world’s most valuable pharma firm. That same drug is now available in India for ₹1,290 a month. It used to cost ₹16,400. That’s a 92% price crash — in three weeks.
And here’s what nobody’s talking about at your trading desk: this is not just a healthcare story. It’s a market story. Let’s break it down.
90%
Price dropin 3 weeks
40+
Indian companiesracing in
$1B
Est. India marketnext 2 years
First — What Even Is This Drug?
Think of it like this. Your body makes a hormone called GLP-1 that tells your brain: “You’re full — stop eating.” The problem is, that signal fades fast. Semaglutide is basically a lab-made version of that same hormone — but it sticks around much longer. Result? You eat less, feel full faster, blood sugar drops, and weight goes down.
Think of it like Paracetamol vs. Dolo. Paracetamol is the molecule. Dolo is the brand name. Same logic — Semaglutide is the molecule, Ozempic is Novo Nordisk’s brand name for it.
The Price War Is Already On
Here’s what happened within 48 hours of patent expiry on March 20, 2026. One event, one morning, and the entire pricing structure of a billion-dollar drug category collapsed:
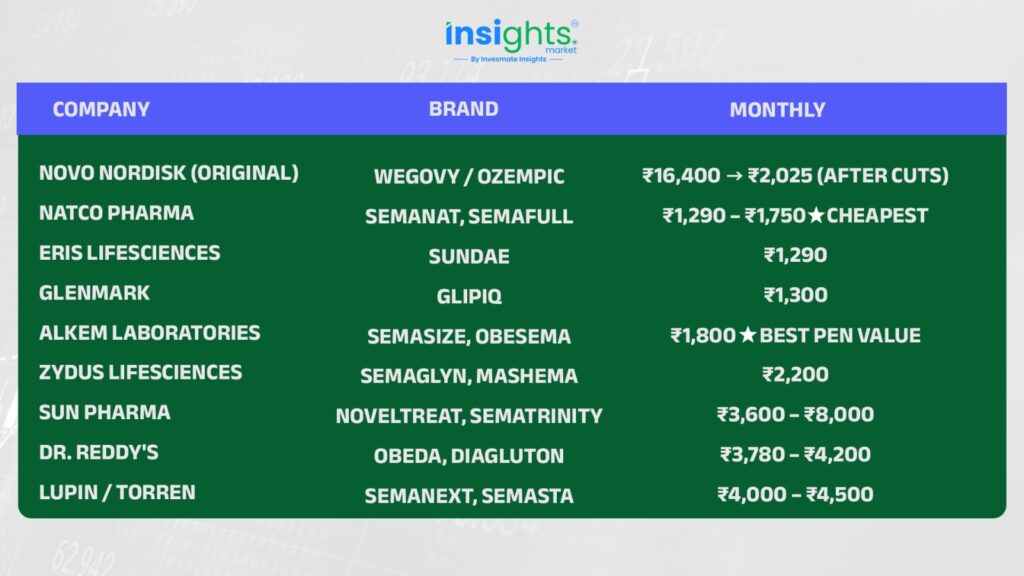
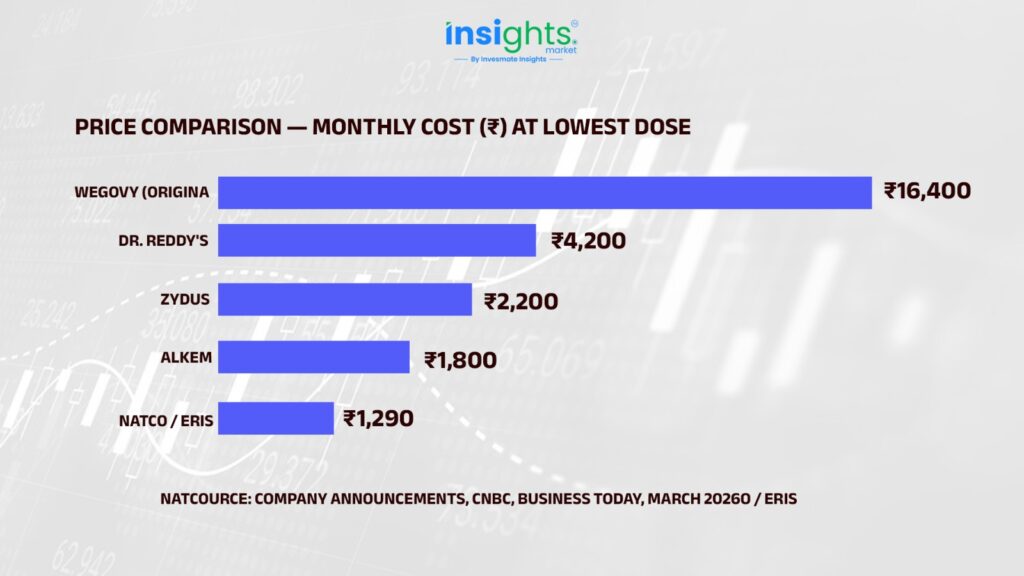
Why did India get generics before the US? India does not grant Patent Term Extensions (PTE/PTA) like the US does. So while America waits until 2032, India’s market opened March 20, 2026 — full stop. Same science, different legal clock.
Where's the Real Investment Opportunity?
Here’s the smarter investor question: instead of betting on which pharma company wins a brutal price war, who profits no matter who wins? These are called proxy plays — and they’re where the real edge is.

The overall market is not small. GLP-1 drug sales in India grew 178% year-on-year in February 2026 alone — before the generic launches even fully played out. Dr. Reddy’s is already targeting 87 countries for export as global patents fall through the 2030s. This is India’s HIV antiretroviral moment — just in a different molecule.
It Goes Beyond Pharma — Your Portfolio Needs to See This
In the US, where 1 in 8 adults is already on a GLP-1 drug, real consumer behaviour has visibly shifted. This is India’s near future — and it affects businesses you’d never connect to a weight-loss injection. The data below is from a Cornell/Numerator study covering the first six months of GLP-1 usage:
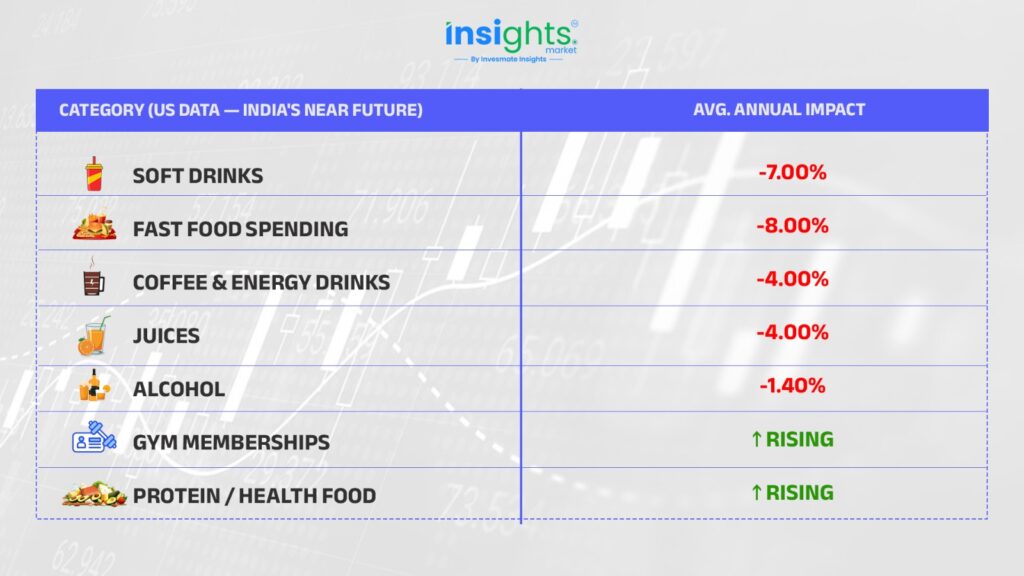
Think about what this means for India — 1.4 billion people, 90 million diabetics, a rapidly growing middle class — and a price barrier that just collapsed overnight. Consumption won’t disappear; it will upgrade. Protein foods, functional nutrition, gym memberships, diagnostics — all beneficiaries of the same trend.
Bottom Line
India is in the very first chapter of this story. The patent just expired. The price just crashed. The market is just opening. In investing, early is exactly where you want to be — once you understand which direction the trend is moving.
A drug changed the world. A patent expiry changed the market. The only question left is: are you paying attention?
FAQs
Dr. Reddy’s Laboratories, Sun Pharmaceutical Industries, Zydus Lifesciences, Lupin Limited, and Alkem Laboratories launched generic semaglutide soon after the March 20, 2026 patent expiry. Among them, Dr. Reddy’s stands out due to its plan to target 80+ global markets as patents expire in other countries.
No. The patent expiry is just the beginning. The Indian semaglutide market could reach about $1 billion in the next two years, and opportunities also extend to diagnostics, medical devices, and digital health platforms.
Ozempic treats Type 2 diabetes, while Wegovy is approved for weight loss. Both are made by Novo Nordisk. Generic semaglutide in India contains the same molecule but costs far less after approval from the Central Drugs Standard Control Organisation.
The US allows Patent Term Extension, which keeps Novo Nordisk’s semaglutide patent valid until 2032. India does not allow this extension, so generics entered the market earlier and reduced prices sharply.
Diagnostics companies like Dr. Lal PathLabs and Metropolis Healthcare, device suppliers like Shaily Engineering Plastics, and digital health platforms such as Tata 1mg could benefit from rising demand.
Margins may be lower, but the huge patient base in India and export opportunities for companies like Dr. Reddy’s and Lupin can still drive strong long-term revenue growth.
This article is for educational and informational purposes only. It is not investment advice or a stock recommendation. Investors should conduct their own research or consult a qualified financial advisor before making investment decisions.




